Company
Beijing Superlaser Technology Co.,Ltd.
Tel
Products
-
Diode Laser Hair Removal Machine
-
Fiber Laser Hair Removal Machine
-
Fractional CO2 Laser Therapy Machine
-
HIFU Wrinkle Removal Machine
-
Q-switch ND YAG laser
-
IPL Machine/PDT Beauty Machine
-
1064 Long Pulse Laser
-
Picosecond Laser
-
Electromagnetic Slim Machine
-
Lipo Laser Slimming Machine
-
Cryolipolysis Body Slimming
-
Vital Injector Mesotherapy Gun
-
Multifunction Skin Care
-
Spare Parts
-
Related Products
808nm Diode Laser Hair Removal Machines Ice Laser Tech FDA & medical CE certificate Medical Laser Device
Brand Name: Boslaser
OEM/ODM: Design on Logo/Appearance/Software/Hardware...
Suitable for: Hospital,Skin Care Center,Beauty Salon...
We're Here to Help :
Easy Ways to Get Answers You Need. 808nm diode laser is one of the main products of Beijing Superlaser Technology Co., Ltd. with good Price. Our company is a professional supplier and manufacturers China, Welcome to buy the best customized system equipment and machines.


Modle HR10 diode laser hair removal machine has medical CE and FDA certificate medical laser device. 808nm wavelength is the Gold standard for hair removal. This machine uses the selective absorption of light to achieve the purpose of hair removal. Because the hair follicles and hair shafts are rich in melanin, The light emitted by the 808nm laser of this machine uses melanin as the target, Melanin absorbs light with a wavelength of 808nm and converts it into heat energy. The temperature of the hair follicle tissue rises sharply, leading to the destruction of hair follicle stem cells, thus achieving the purpose of removing hair.
808nm diode laser is one of the main products of Beijing Superlaser Technology Co., Ltd. with good Price. Our company is a professional supplier and manufacturers China, Welcome to buy the best customized system equipment and machines.
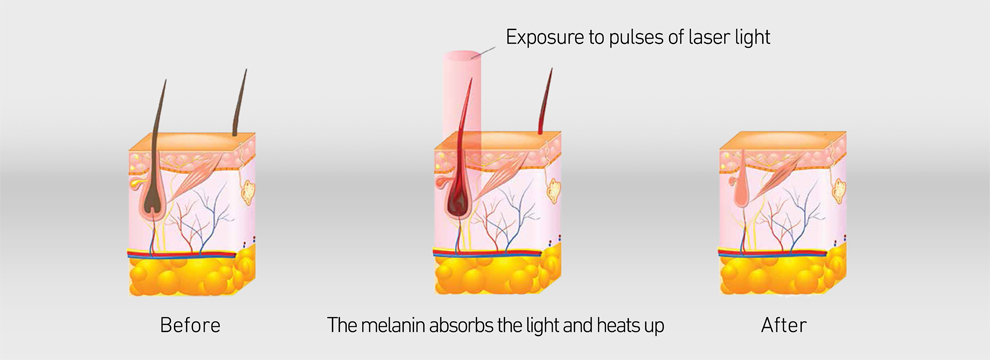

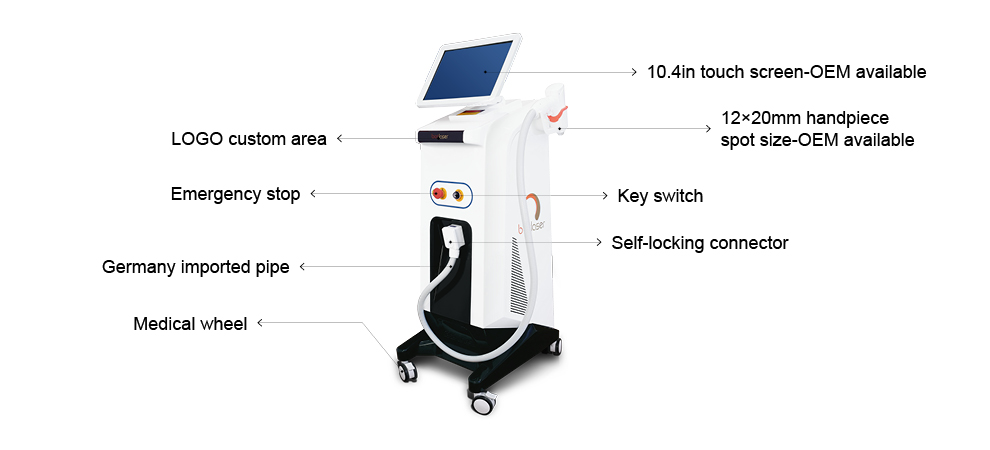
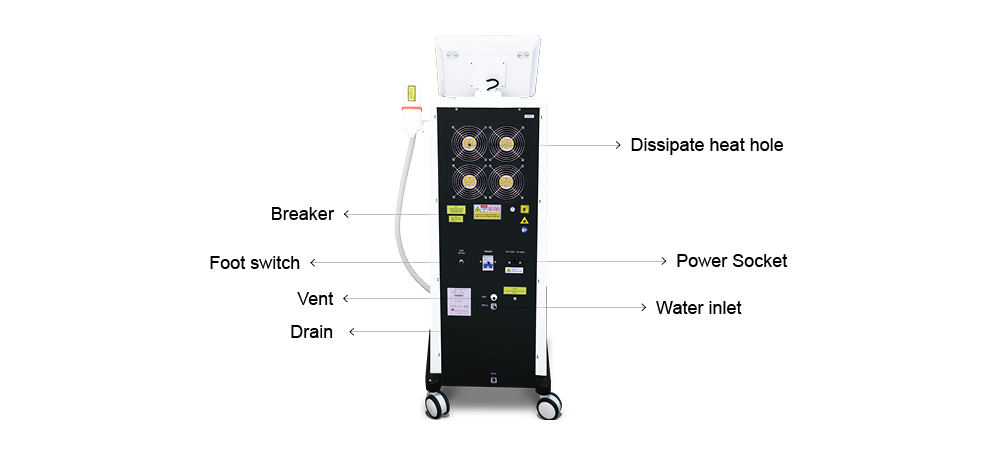

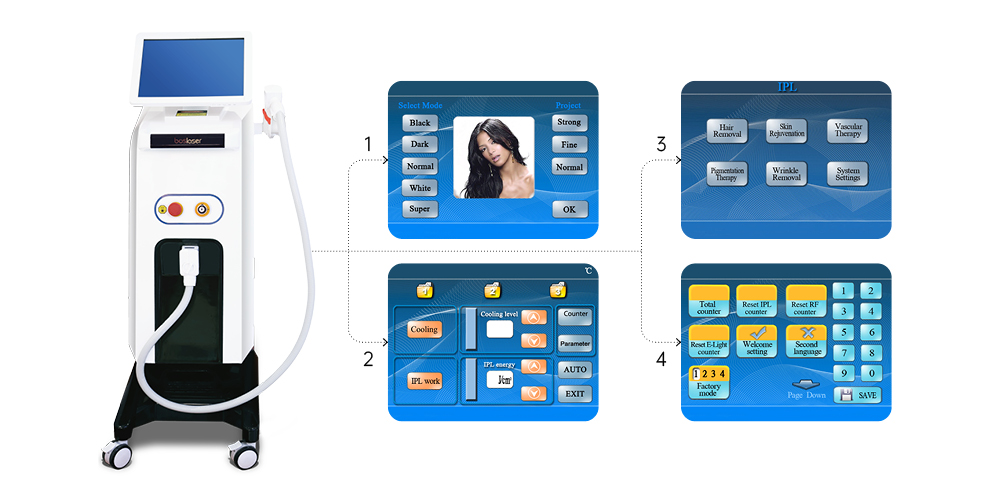

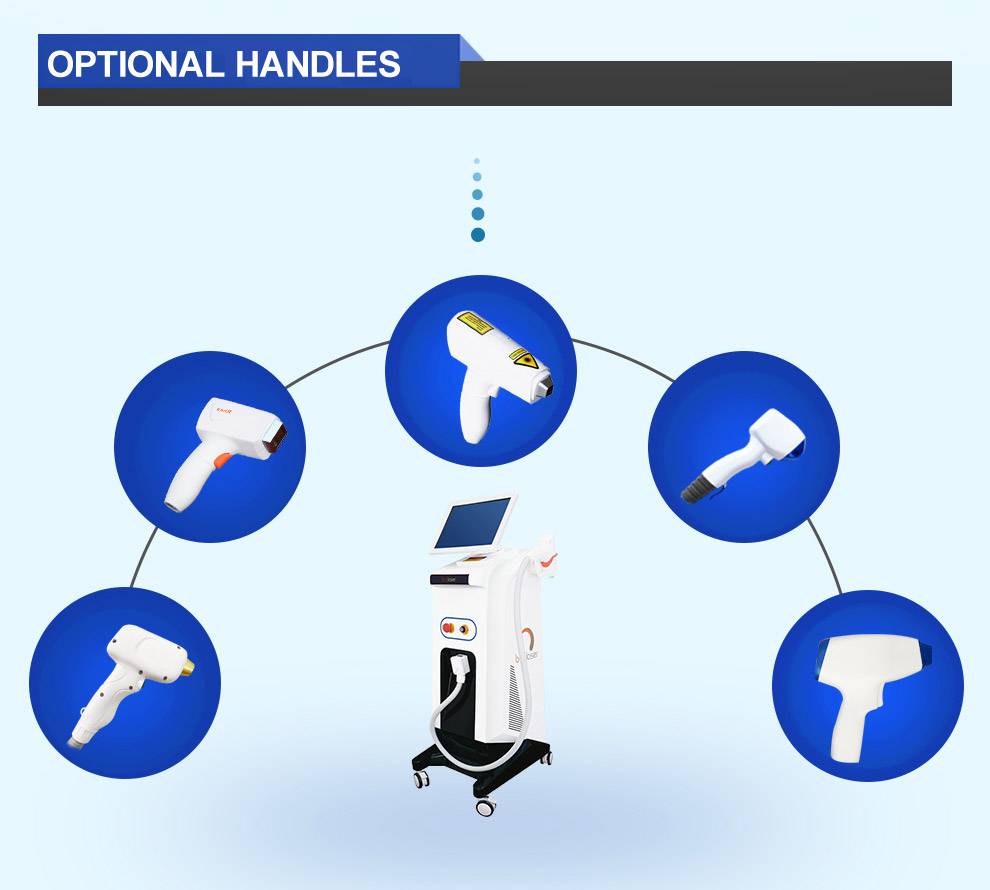
This medical laser device laser hair removal machines handle has a high power : 300W/600W/1000W/1200W...which support OEM and ODM, and the laser bar is imported from U.S. , it is cost-effective and higher quality. The laser hair removal machine' handle also has intelligent temperaturer monitoring system, the tempertature sensor is installed in the handle which can keep the temperature of the Sapphire contact cooling system between 0°C and -4°C to ensure safe and comfortable treatment.

Energy Test


1. Hair Removal, laser epilation, Skin Tightening;
2. Widely used for any color hair;
3. Widely used for any skin type;
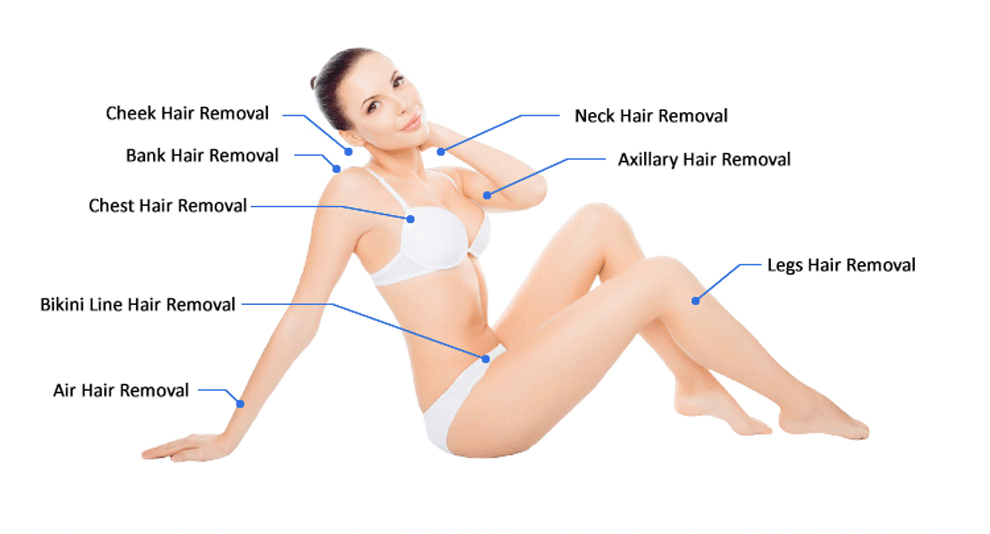
4. Removel Area: Lip Hair, Beard, Chest Hair, Armpit Hair, Back Hair, Arm Hair, Leg Hair, Unwanted Hair outside the Bikini Line.


1.Stable high output power diode laser bars.
2.More than 20,000,000 shots ensure longer life time than other hair removal machine.
3.Strong Sapphire contact cooling system laser hair removal machines
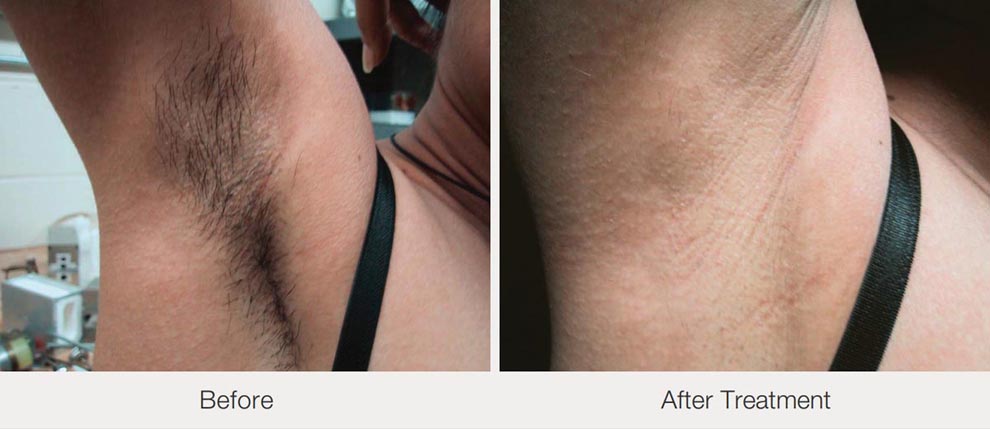
4.Excellent treatment result can be expected at the first treatment and suitable to all types of hair
5.Safety: Normal skin tissue absorbs less 808NM laser light, and hair follicles absorb more melanin, ensuring that all energy is applied to hair follicles. At the same time, the skin tissue around the hair follicle is intact.
6.Comfort: The TEC constant temperature refrigeration system ensures that the sapphire light outlet is kept at a constant temperature of -4°C to achieve painless hair removal.
7.Fast: 12*10CM super large spot + 10HZ light frequency greatly improves the treatment speed and saves treatment time.
8.Curative effect: The high energy density output of the coherent imported laser ensures that the laser energy acting on the hair is stronger and the effect is better.

|
Light Source |
Diode Array with collimator Lens |
Screen |
10.4 inch true color touch screen LCD |
|
Modes |
Single Pulse |
Window Spot Size |
12 x 10mm = 1.2cm2 |
|
Wavelength |
808nm |
Pulse Repetition |
Up to 20 pulses per second |
|
Output Energy |
Up to 120J/cm2 |
Tip Cooling |
Sapphire Peltier contact cooling |
|
Power requirement |
110V 60 Hz or 230V 50Hz |
System Cooling |
Compressor closed circuit water chiller with integrated heat exchanger.Inner distilled water circulation cooling system & air cool- ing ( maximum water pressure: 0.3MPa ) |
|
Rated Power |
1500VA |
||
|
Temperature |
20°C—25°C |
Dimensions |
598L x 440W x 1093H |
|
Relative humidity |
≤70% |
Laser Class |
Class 4 Laser Product |
|
Pressure |
860—1060hpa |
Weight |
60 kilogram |



★ LEAVE A MESSAGE
Beijing Super laser Technology Co., Ltd.

Beijing Superlaser Technology Co., Ltd
Professional Aesthetics & Medical Laser Equipment Manufacturer. FDA/Medical CE/TUV / ISO13485 Approved. 14-Year Experience in OEM/ODM Service.
2 floor,building3, No.2 zhongfu street,
xihongmen town,daxing district,Beijing
Newsletter
Copyright © Beijing Super laser Technology Co., Ltd. All rights reserved.
京ICP备10032915号
Powered By:
www.300.cn

Send message
Please fill out the form below and we will get back to you as soon as possible.






